Unit #2, Matter & Chemical Change
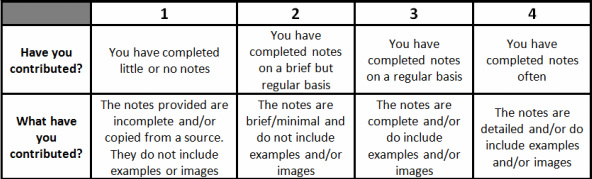
Science notes rubric:
Topic #1
The following concepts are important from the topic, make sure that you have them in your notes!
1. The definition of chemistry
2. WHIMIS symbols and lab safety including the definition of caustic
3. The particle model and how it works
4. The different types of mixtures and pure substances
1. The definition of chemistry
2. WHIMIS symbols and lab safety including the definition of caustic
3. The particle model and how it works
4. The different types of mixtures and pure substances
Topic #2
The following concepts are important from the topic, make sure that you have them all in your notes!
5. The definition of a physical change
6. The definition of a chemical change
7. The 7 observations of a chemical change
8. Physical properties
9. Chemical properties
10. Qualitative and Quantitative properties
5. The definition of a physical change
6. The definition of a chemical change
7. The 7 observations of a chemical change
8. Physical properties
9. Chemical properties
10. Qualitative and Quantitative properties
|
|
Topic #3
The following concepts are important from the topic, make sure that you have them in all your notes!
11. The Law of Conservation of Mass, be able to explain what it is and how it works
12. The Law of Definite Composition, be able to explain what it is and how it works
13. Be able to state and explain what electrolysis is and how it works
14. Be able to list and explain Daltons Atomic Theory
15. Be able to explain what an atom is and how it works, including all the different parts and their definitions. (proton, electron, neutron, atomic nucleus)
16. Know and be able to explain the difference between an element and a compound
17. Know what laws and theories are and the differences between them. Also know the scientific process of how observations can become theories
11. The Law of Conservation of Mass, be able to explain what it is and how it works
12. The Law of Definite Composition, be able to explain what it is and how it works
13. Be able to state and explain what electrolysis is and how it works
14. Be able to list and explain Daltons Atomic Theory
15. Be able to explain what an atom is and how it works, including all the different parts and their definitions. (proton, electron, neutron, atomic nucleus)
16. Know and be able to explain the difference between an element and a compound
17. Know what laws and theories are and the differences between them. Also know the scientific process of how observations can become theories
|
|
Topic #4
The following concepts are important from the topic, make sure that you have them all in your notes!
18. How to read the periodic table, know the first 18 periodic symbols, their elements name and number
19. Know where the metals, metaliods and non-metals are on the periodic table, along with their common properties
20. Know the different chemical families, where they are on the periodic table and their common properties and how reactive they are (alkali metals, alkali earth metals, noble gases and halogens)
21. Be able to explain how reactive an element is depending on the number of electrons it has.
18. How to read the periodic table, know the first 18 periodic symbols, their elements name and number
19. Know where the metals, metaliods and non-metals are on the periodic table, along with their common properties
20. Know the different chemical families, where they are on the periodic table and their common properties and how reactive they are (alkali metals, alkali earth metals, noble gases and halogens)
21. Be able to explain how reactive an element is depending on the number of electrons it has.
Topic #5
The following concepts are important from the topic, make sure that you have them all in your notes!
22. Who is Dimitri Mendeleev and how did he build the periodic table of elements
23. How do you use the peridoic table to find the following information for each element: Atomic Mass, Number of Protons, Electrons & Neutrons
22. Who is Dimitri Mendeleev and how did he build the periodic table of elements
23. How do you use the peridoic table to find the following information for each element: Atomic Mass, Number of Protons, Electrons & Neutrons
Topic #6
The following concepts are important from the topic, make sure that you have them all in your notes!
24. What is a molecular compound and how does it work?
25. What is an ionic compound and how does it work?
26. What is a chemical formula and how do they work?
27. What is a diatomic molecule
28. What are characteristics of molecular compounds?
29. What are characteristics of ionic compounds?
30. How are molecular compounds named?
31. How are ionic compounds named?
24. What is a molecular compound and how does it work?
25. What is an ionic compound and how does it work?
26. What is a chemical formula and how do they work?
27. What is a diatomic molecule
28. What are characteristics of molecular compounds?
29. What are characteristics of ionic compounds?
30. How are molecular compounds named?
31. How are ionic compounds named?
|
|
|
|
Topic #7
The following concepts are important from the topic, Make sure that you have them in all your notes!
32. How do you write a chemical reaction properly? Where and how do you list the reactants and the products?
33. How do you change a chemical equation to a word equation?
34. In a chemical reaction what do the numbers and subscripts mean?
35. What does it mean if a reaction is exothermic or endothermic?
32. How do you write a chemical reaction properly? Where and how do you list the reactants and the products?
33. How do you change a chemical equation to a word equation?
34. In a chemical reaction what do the numbers and subscripts mean?
35. What does it mean if a reaction is exothermic or endothermic?
|
|
Topic #8
The following concepts are important from the topic, make sure that you have them in all your notes!
36. What is the reaction rate for a chemical reaction, how does surface area, temperature and agitation affect it?
37. What is a catalyst and how does it work
38. What is an enzyme and how does it work?
39. What is an inhibitor and how does it work
40. Be familair with the chemical reaction know as corrosion and how to prevent it
41. Be familiar with the chemical reaction know as combustion along with it's reactants and products
36. What is the reaction rate for a chemical reaction, how does surface area, temperature and agitation affect it?
37. What is a catalyst and how does it work
38. What is an enzyme and how does it work?
39. What is an inhibitor and how does it work
40. Be familair with the chemical reaction know as corrosion and how to prevent it
41. Be familiar with the chemical reaction know as combustion along with it's reactants and products