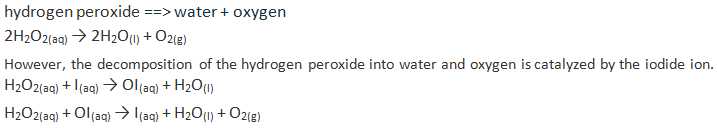
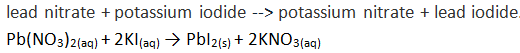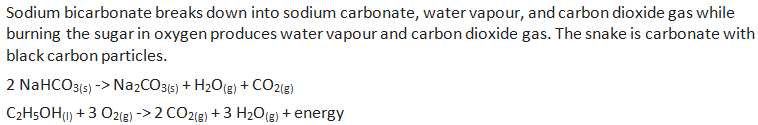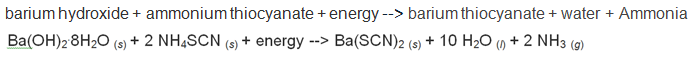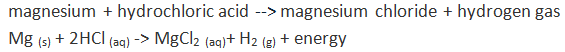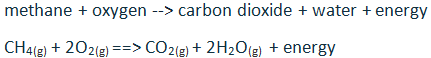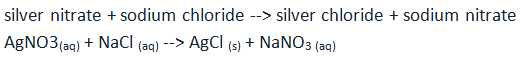Unit #2 Project, Chemical Reaction Research & Performance
Your task will be to research and demonstration one of the following chemical reactions. For your demonstration/reaction you will need to provide the following information:
1. - What elements are involved
2. - What types of compounds are involved, would these compounds conduct electricity on their own?
3. - Is your reaction exothermic, endothermic or neither?
4. - What states are each of the reactants and products in?
5. - What observations can you make to prove that a chemical reaction has occurred?
6. - What is qualitative and quantitative data/observations for your reaction
7. - What could be done to speed or slow down the reaction rate of your chemical reaction?
8. - Explain the law of conservation of mass using your reaction
9. - Explain the law of definite composition using compounds within your reaction.
10. - Show many electrons, protons or neutrons the elements within your reaction should they have?
2. - What types of compounds are involved, would these compounds conduct electricity on their own?
3. - Is your reaction exothermic, endothermic or neither?
4. - What states are each of the reactants and products in?
5. - What observations can you make to prove that a chemical reaction has occurred?
6. - What is qualitative and quantitative data/observations for your reaction
7. - What could be done to speed or slow down the reaction rate of your chemical reaction?
8. - Explain the law of conservation of mass using your reaction
9. - Explain the law of definite composition using compounds within your reaction.
10. - Show many electrons, protons or neutrons the elements within your reaction should they have?